Second-Semester Mathematics
Our Chemistry and Math teachers swap jobs every semester. So: first semester, Laura Webb was my Math teacher and Dr. Don Mackay was my Chemistry teacher. Now, Second Semester, Laura is my Chemistry teacher and Dr. Don is my Math teacher.
In the Dr. Don Math class, we participated in a multi-discipline Green House Gas project. The final report of this project is below:
In the Dr. Don Math class, we participated in a multi-discipline Green House Gas project. The final report of this project is below:
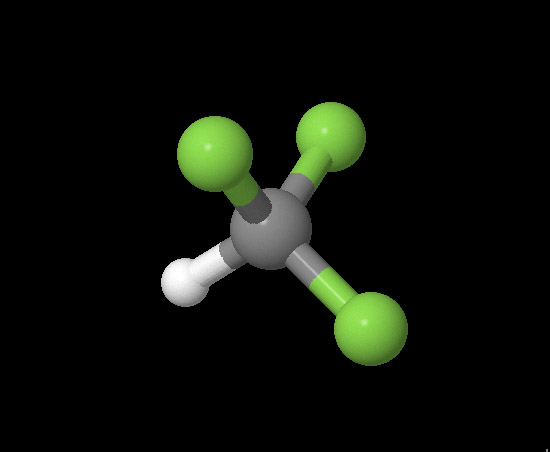
Fluoroform (CHF3) Version 1: Greenhouse Gas?
Background:
|
This project was structured around the understanding of how greenhouse gases function in our world, and what they actually are on an atomic level. We studied "the greenhouse effect" (what greenhouse gases do and how they do it), and then applied that information to a new gas to see if it fit the bill.
The greenhouse effect describes what happens to light that enters the Earth'e atmosphere. 49% of all light is scattered and reflected by air molecules, clouds, and the surface of the earth. However, that leaves 51% of the light photons emitted by the sun to be absorbed by the earth. When the earth re-emitts the photons, it is not in the same form the earth received them in (our planet does not glow), but as infrared radiation (IR). Most of IR is absorbed by greenhouse gases Earth's atmosphere, which is warmed by the contact. So what are greenhouse gases, and how can their contact with IR cause the warming of Earth's atmosphere? There are several factors: Firstly, the molecule must have at least 3 atoms in it, so it can vibrate in an oscillating dipole pattern (where the center of charge moves, upending the molecule's balanced charges and creating a dipole). If an IR wave comes into contact with such a molecule, then it can be absorbed. When atoms absorb photons, the atom's electrons jump to orbitals further from the center and the entire molecule speeds up with energy. Secondly, in order for the IR wave to be absorbed, the molecule must resonate (vibrate) at the same frequency as the IR. Thirdly, the now "excited" molecule must move faster, producing heat energy in response to the added energy of the IR photons. I chose the molecule Fluoroform to work with in this project (picture and destription are at right). I hypothesize: If Fluoroform can change its dipole moment when a scale factor is introduced, then Fluoroform is a greenhouse gas because it would then be conceivably possible for Fluoroform to exist at different frequencies, including the frequency of IR light. I will test this hypothesis using a dipole moment calculator. |
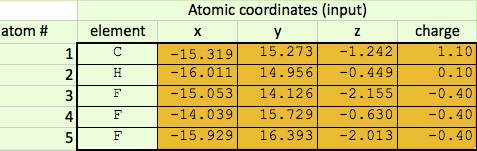
Input:
Procedure:
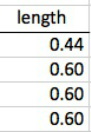
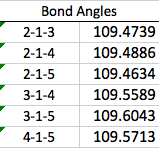
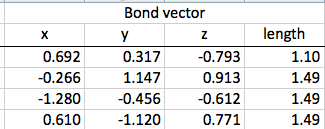
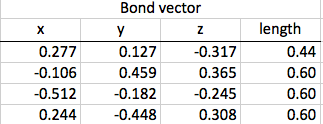
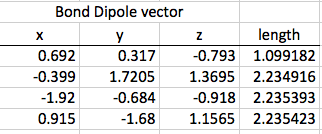
The first step of this calculation process is to find the differences between the bond vectors. This consists of subtracting one from another. Once the differences in the x, y, and z coordinates are all found, the distance formula can be used to find the final length of the vector.
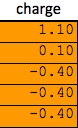
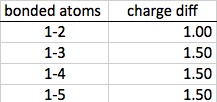
Next, the charge differences are found between the various atoms, and these differences are multiplies by the applicable bond vector found on step one. Again, once all of the x, y, and z coordinates are found for a particular pair of atoms, those figures can be combined using the distance formula.
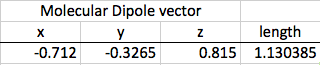
Thirdly, I can use this information to find the dipole moment of the entire molecule. I add all the x values together, all the y values, and all the z values to find the final coordinates, which I can input into another distance formula to find the final dipole moment.
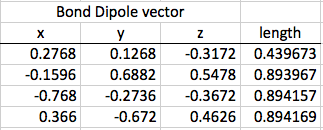
Now that I have calculated the original dipole moment of my molecule, I can distort it using a scale factor to see if the dipole moment changes. I used a symmetric stretch: I multiplied all of the coordinates in the first step by 0.4, and then proceed through the rest of steps one, two, and three with that change.
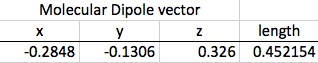
The original dipole moment for my molecule was 1.13. The distorted dipole moment was 0.452.
Next, the charge differences are found between the various atoms, and these differences are multiplies by the applicable bond vector found on step one. Again, once all of the x, y, and z coordinates are found for a particular pair of atoms, those figures can be combined using the distance formula.
Thirdly, I can use this information to find the dipole moment of the entire molecule. I add all the x values together, all the y values, and all the z values to find the final coordinates, which I can input into another distance formula to find the final dipole moment.
Now that I have calculated the original dipole moment of my molecule, I can distort it using a scale factor to see if the dipole moment changes. I used a symmetric stretch: I multiplied all of the coordinates in the first step by 0.4, and then proceed through the rest of steps one, two, and three with that change.
The original dipole moment for my molecule was 1.13. The distorted dipole moment was 0.452.
Results:
Conclusion:
So, in the end, is Fluoroform a greenhouse gas? A look back at the hypothesis (If Fluoroform can change its dipole moment when a scale factor is introduced, then Fluoroform is a greenhouse gas because it would then be conceivably possible for Fluoroform to exist at different frequencies, including the frequency of IR light.) and a look at my results (The original dipole moment for my molecule was 1.13. The distorted dipole moment was 0.452.) makes the answer very clear: Yes, Fluoroform is a greenhouse gas. It has the capacity to absorb IR and convert the light photon into energy, warming the atmosphere.